|
18.07.2022 13:45:00
|
RENUVION® CLEARED BY FDA FOR NECK LAXITY PROCEDURES OFFERING PATIENTS A MINIMALLY INVASIVE OPTION TO ELIMINATE SAGGING NECK
Exclusive Apyx Medical Device Delivers State-Of-The-Art Plasma Energy To Improve Appearance of Loose Skin on The Neck and Chin Available for Physicians and Patients
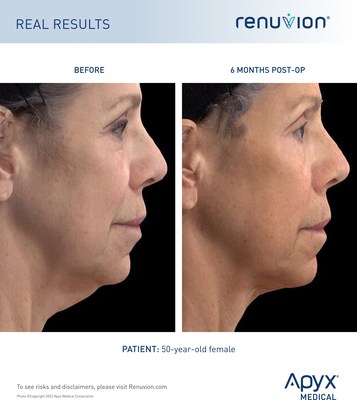
CLEARWATER, Fla., July 18, 2022 /PRNewswire/ -- Apyx Medical Corporation (NASDAQ: APYX), the manufacturer of Renuvion®, a proprietary helium plasma and radiofrequency technology, announces the receipt of 510(k) clearance from the U.S. Food and Drug Administration ("FDA") for the use of Renuvion for use in subcutaneous dermatological and aesthetic procedures to improve the appearance of lax skin in the neck and submental region. This clearance offers a new option for physicians and patients to improve loose skin on the neck and submental region.
With this clearance Renuvion is the only product that is FDA-cleared to improve the appearance of loose skin on the neck and chin. Renuvion's patented technology offers a game-changing option in cosmetic surgery for neck laxity procedures.
Loss of jawline definition with age is inevitable and can add years to one's appearance. Renuvion provides a minimally-invasive and cost-effective option with minimal downtime. The results reveal a more contoured and smoother neck and jawline.
Within the fat layer beneath the skin, there is a web-like structure of collagen fibers. These fibers provide support and elasticity to the skin itself. Age, weight fluctuations, and sun exposure contribute to the breakdown of these fibers, causing the skin to sag. Surface treatments often can't delivery energy deep enough to effectively treat these fibers, and invasive surgery can be painful and is associated with having a long downtime. The Renuvion energy is used in a minimally-invasive procedure to directly treat the collagen fibers and address the root cause of neck and chin laxity.
"We are very pleased to receive FDA clearance enabling Apyx Medical's Renuvion technology to be used in neck and chin laxity procedures," said Charlie Goodwin, President and Chief Executive Officer. "We've seen a considerable uptick in the market for these cosmetic procedures and are thrilled to be able to offer our unique technology to physicians and their patients to meet this growing demand – it's the next generation option for neck contouring procedures."
For a list of physicians, by state, offering Renuvion in their practice visit https://RenuvionFinder.com.
For more information visit Renuvion.com.
About Apyx Medical Corporation:Apyx Medical Corporation is an advanced energy technology company with a passion for elevating people's lives through innovative products in the cosmetic and surgical markets. Known for its innovative Helium Plasma Technology, Apyx is solely focused on bringing transformative solutions to the physicians and patients it serves. The company's Helium Plasma Technology is marketed and sold as Renuvion® in the cosmetic surgery market and J-Plasma® in the hospital surgical market. Renuvion® offers surgeons and physicians a unique ability to provide controlled heat to the tissue to achieve their desired results. The J-Plasma® system allows surgeons to operate with a high level of precision while minimizing unintended tissue trauma. The Company also leverages its deep expertise and decades of experience in unique waveforms through original equipment manufacturing (OEM) agreements with other medical device manufacturers. For further information about the Company and its products, please refer to the Apyx Medical Corporation website at www.ApyxMedical.com.
Cautionary Statement on Forward-Looking StatementsCertain matters discussed in this release and oral statements made from time to time by representatives of the Company may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Although the Company believes that the expectations reflected in such forward-looking statements are based upon reasonable assumptions, it can give no assurance that its expectations will be achieved.
All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including but not limited to, any statements regarding the potential impact of the COVID-19 pandemic and the actions by governments, businesses and individuals in response to the situation; projections of net revenue, margins, expenses, net earnings, net earnings per share, or other financial items; projections or assumptions concerning the possible receipt by the Company of any regulatory approvals from any government agency or instrumentality including but not limited to the U.S. Food and Drug Administration, supply chain disruptions, component shortages, manufacturing disruptions or logistics challenges; or macroeconomic or geopolitical matters and the impact of those matters on the Company's financial performance.
Forward-looking statements and information are subject to certain risks, trends and uncertainties that could cause actual results to differ materially from those projected. Many of these factors are beyond the Company's ability to control or predict. Important factors that may cause the Company's actual results to differ materially and that could impact the Company and the
statements contained in this release include but are not limited to risks, uncertainties and assumptions relating to the regulatory environment in which the Company is subject to, including the Company's ability to gain requisite approvals for its products from the U.S. Food and Drug Administration and other governmental and regulatory bodies, both domestically and internationally; the impact of the recent FDA Safety Communication on our business and operations; factors relating to the effects of the COVID-19 pandemic; sudden or extreme volatility in commodity prices and availability, including supply chain disruptions; changes in general economic, business or demographic conditions or trends; changes in and effects of the geopolitical environment; liabilities and costs which the Company may incur from pending or threatened litigations, claims, disputes or investigations; and other risks that are described in the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2021 and the Company's other filings with the Securities and Exchange Commission. For forward-looking statements in this release, the Company claims the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995. The Company assumes no obligation to update or supplement any forward-looking statements whether as a result of new information, future events or otherwise.
Media Contact:
Jane Sparango
Coterie Media (for Renuvion)
jane@coteriemedia.com
310-339-1214
Investor Relations Contact:
ICR Westwicke on behalf of Apyx Medical Corporation
Mike Piccinino, CFA
investor.relations@apyxmedical.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/renuvion-cleared-by-fda-for-neck-laxity-procedures-offering-patients-a-minimally-invasive-option-to-eliminate-sagging-neck-301587982.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/renuvion-cleared-by-fda-for-neck-laxity-procedures-offering-patients-a-minimally-invasive-option-to-eliminate-sagging-neck-301587982.html
SOURCE Renuvion
 Der finanzen.at Ratgeber für Aktien!
Der finanzen.at Ratgeber für Aktien!
Wenn Sie mehr über das Thema Aktien erfahren wollen, finden Sie in unserem Ratgeber viele interessante Artikel dazu!
Jetzt informieren!
Nachrichten zu Bovie Medical Corp Registered Shsmehr Nachrichten
|
07.11.24 |
Ausblick: Bovie Medical vermeldet Zahlen zum jüngsten Quartal (finanzen.net) | |
|
07.08.24 |
Ausblick: Bovie Medical legt Zahlen zum jüngsten Quartal vor (finanzen.net) |
Analysen zu Bovie Medical Corp Registered Shsmehr Analysen
Aktien in diesem Artikel
| Bovie Medical Corp Registered Shs | 1,37 | 4,58% |
|